Updated on 18 July 2024 by: Yeo Yu Teng
What Is Glycerine | Types of Glycerine | How Is It Made | Uses of Glycerine | Is It Safe | Palm Oil to Refined Glycerine | Largest Glycerine Refining Unit
Glycerine, also referred to as glycerol, is a clear, odorless liquid commonly used in food, skincare, pharmaceuticals, and industrial products. Although it is sometimes mistaken for nitroglycerin, glycerine is entirely distinct and safe for everyday use.
What exactly is glycerine, how is it produced, and why is it such a versatile ingredient? In this guide, we will explore its benefits, common applications across various industries, and the production process behind this essential compound.
What Is Glycerine?
Glycerine is a natural compound sourced from vegetable oils or animal fats. Despite its origin in fats, it is not oily in nature.
It is a colorless, clear liquid with a syrupy consistency. While glycerine does not have a distinct odor, it has a notably sweet taste.
For science enthusiasts, glycerine’s chemical formula is C3H8O3. Its chemical structure classifies it as a sugar alcohol or polyol, though it does not have intoxicating effects like the ethanol found in wines and beers.
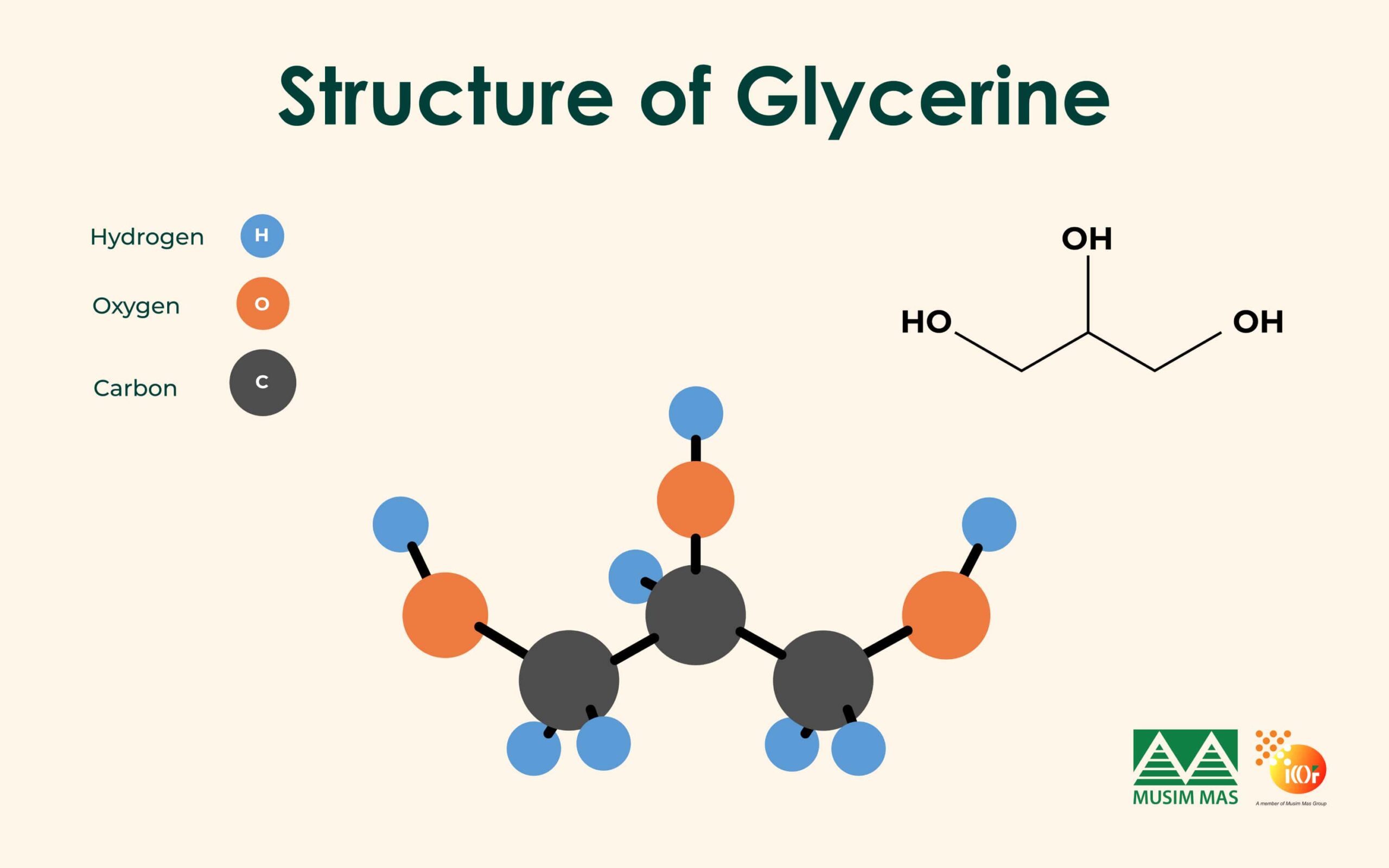
One of the key characteristics of glycerine is its ability to function as a humectant, meaning it attracts water from its surroundings.
This property, resulting from the three hydroxyl (-OH) groups in its structure, makes glycerine an effective moisturizer and preservative.
The names glycerine or glycerol may cause confusion, as the “ine” suffix often implies an amine, which contains the -NH2 functional group. The term glycerol is more accurate, as the “ol” suffix signifies an alcohol.
Read More: Vegetable Glycerin for Innovations in Low-Sugar Food and Beverages
Types of Glycerine
Glycerine typically exists in two primary forms: natural and synthetic. Below are the explanations for each type.
Natural Glycerine
Natural glycerine is derived from plant oils or animal fats. Vegetable-based glycerine often comes from palm oil, coconut oil, soy, or rapeseed. This type is commonly used in food, cosmetics, and pharmaceuticals, and is suitable for vegan products.
Animal-based glycerine is usually obtained from beef or mutton fat. It has the same chemical structure as vegetable glycerine but may not meet vegan or vegetarian standards.
Synthetic Glycerine
Synthetic glycerine is made from petroleum-based propylene. Although its origin differs, its chemical composition is identical to natural glycerine.
It is sometimes confused with propylene glycol. However, propylene glycol contains only two hydroxyl groups, while glycerine contains three, which affects how each behaves in formulations.
How Is Glycerine Made?
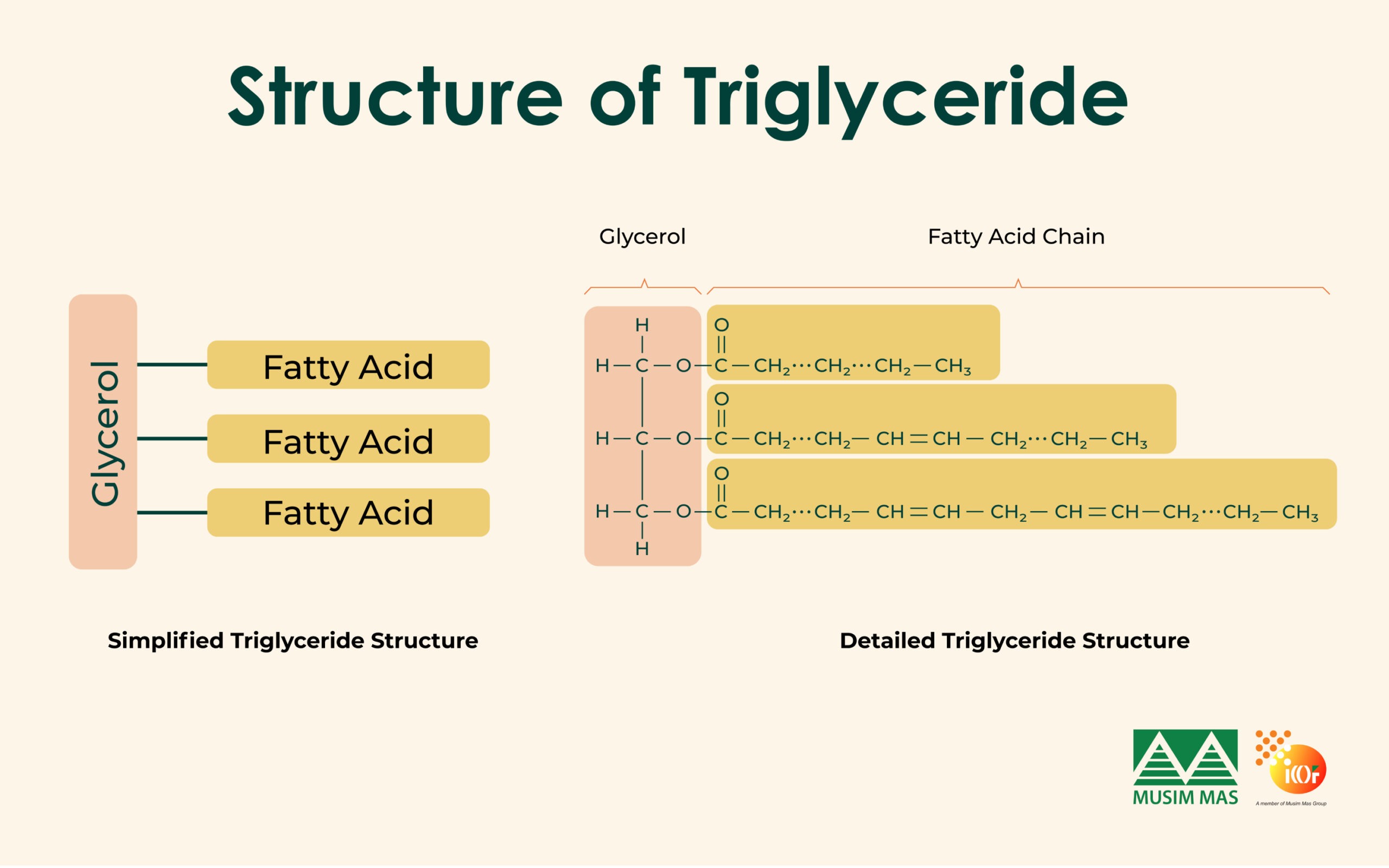
Glycerine is produced from triglycerides, which are the main components of fats and vegetable oils. Triglycerides consist of three fatty acid chains attached to one glycerol molecule. To obtain glycerine, the bond between the fatty acids and glycerol must be broken.
Glycerine was first discovered in 1783 by Swedish chemist Carl Wilhelm Scheele. Today, it is primarily produced through three industrial processes.
A Swedish chemist named Carl Wilhelm Scheele was heating olive oil together with another chemical containing lead when he accidentally discovered glycerine. He then reported his findings and the glycerine production process in 1783.
Today, we can produce crude glycerine in three ways: hydrolysis, saponification, and transesterification. Crude glycerine is then distilled and bleached in distillation plants to refined glycerine.

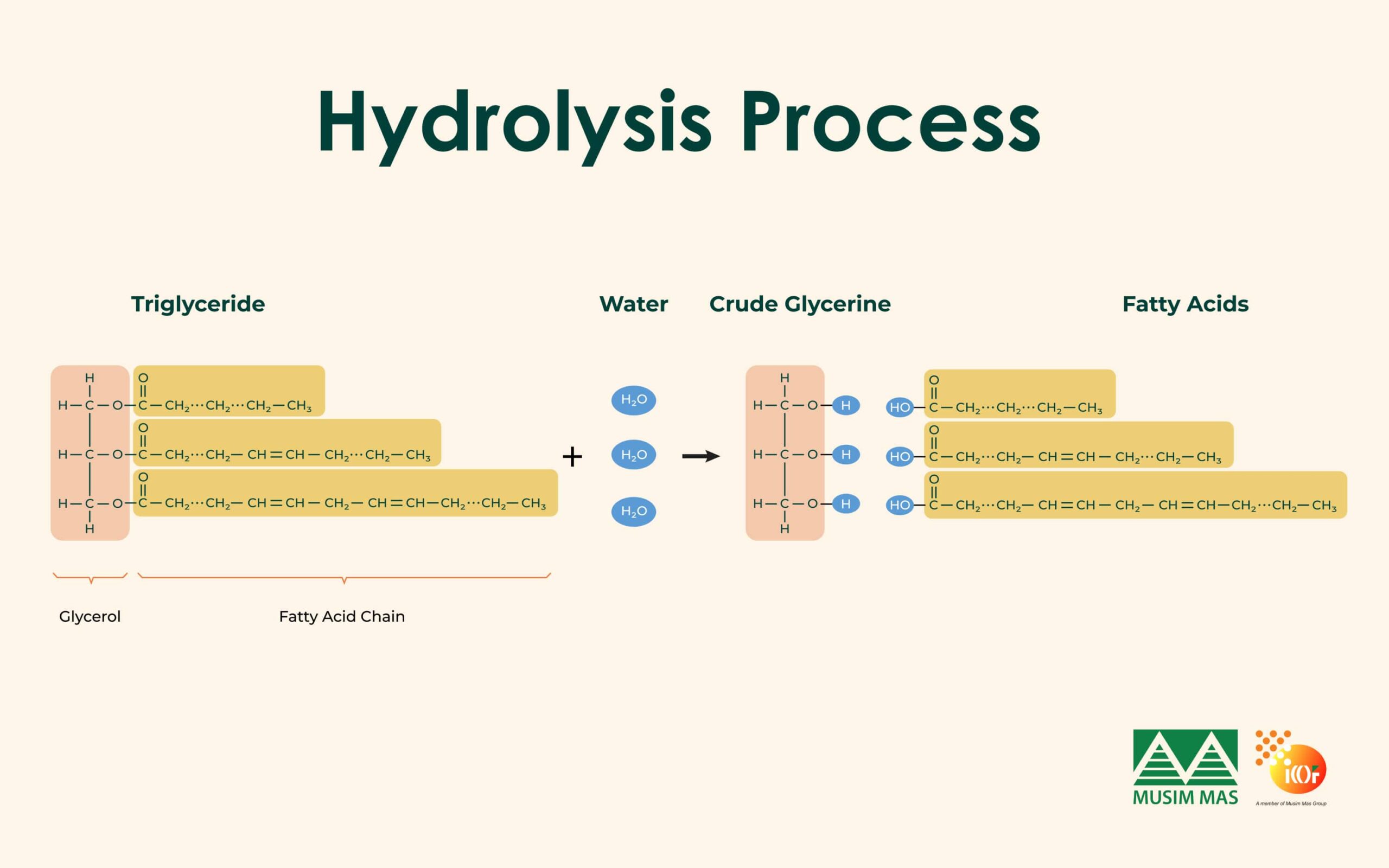
Hydrolysis
Hydrolysis involves heating oils or fats with water under high pressure. This breaks the bonds between fatty acids and glycerol, producing free fatty acids and crude glycerine.
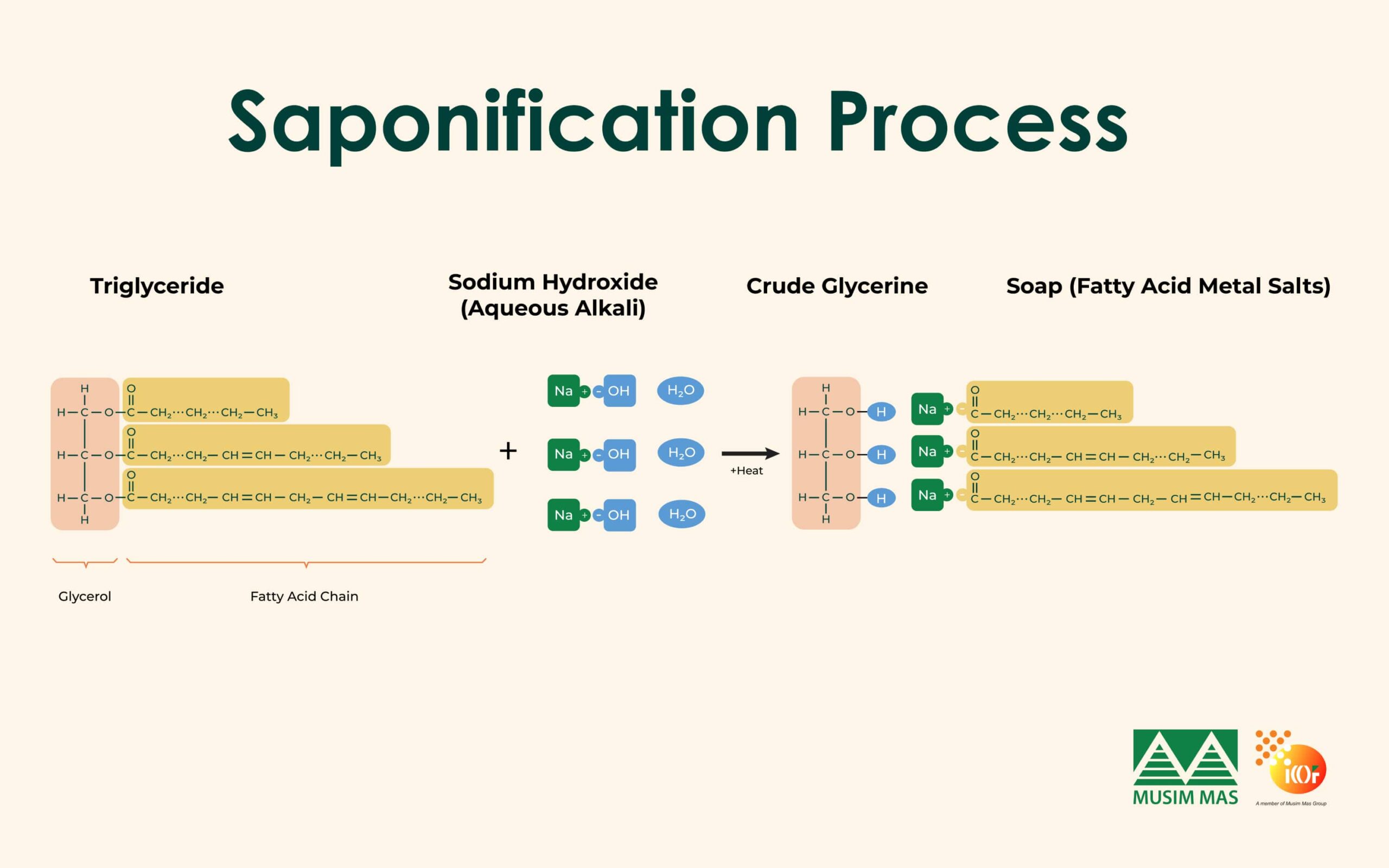
Saponification
Saponification is commonly known as soap-making. Fats are heated with an alkaline solution such as sodium hydroxide. This process splits fats into soap and crude glycerine. Historically, this was one of the main ways glycerine was produced.
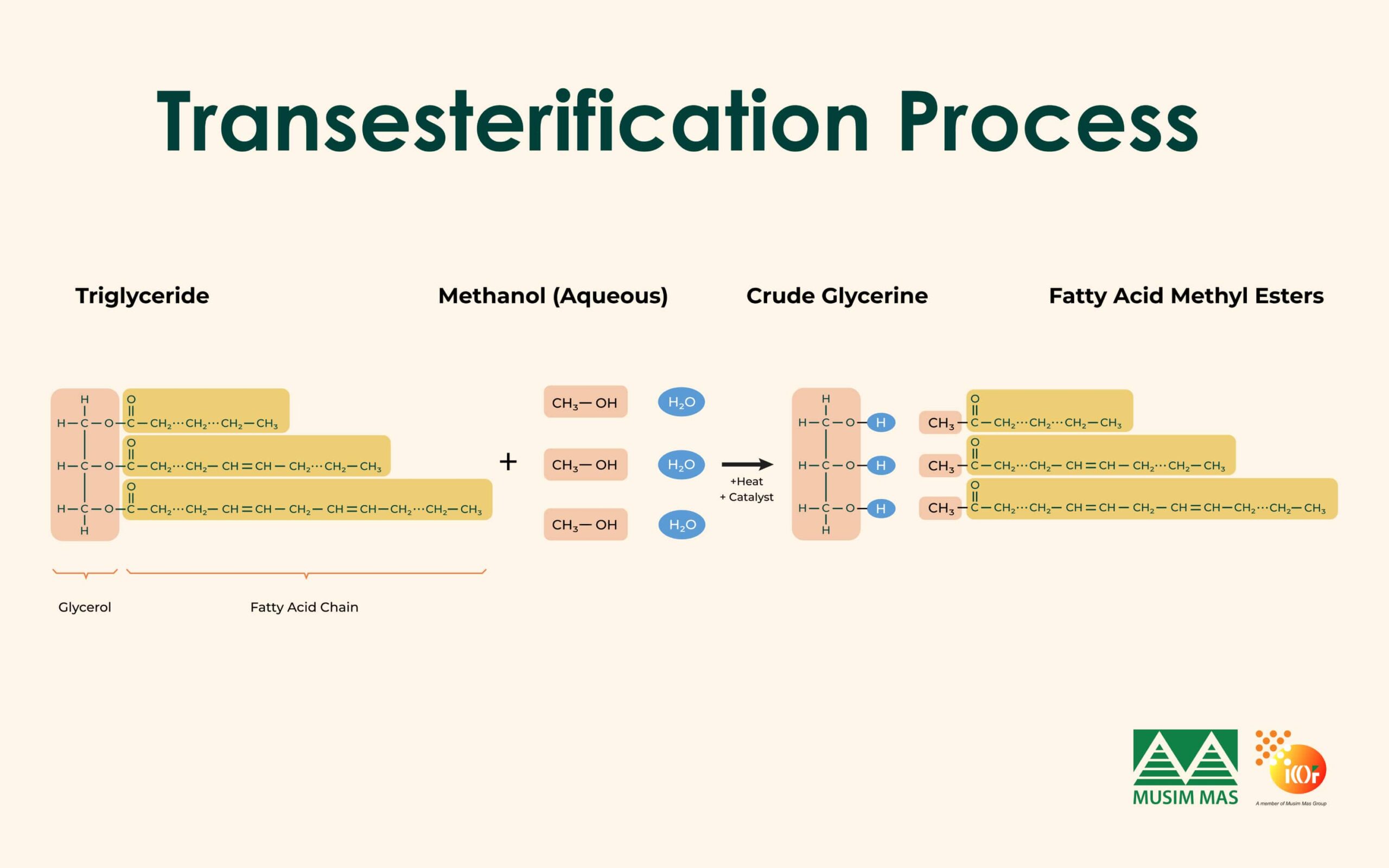
Transesterification
Today, the majority of glycerine is produced as a by-product of biodiesel production. In this process, oils or fats react with alcohol, usually methanol, in the presence of a catalyst. The reaction produces biodiesel and crude glycerine as a by-product.
As a result, a significant portion of the global glycerine supply is derived from biodiesel manufacturing.
Refining Crude Glycerine
Crude glycerine contains impurities such as water, salts, and residual fatty acids. It undergoes distillation and bleaching to produce refined glycerine with high purity levels suitable for food, pharmaceutical, and cosmetic use.
What Is Glycerine Used For?
Glycerine is highly valued for its versatility, finding applications across numerous industries due to its unique chemical properties. Below are some of the primary uses of glycerine:
Food and Beverage Applications
In food production, glycerine serves several functions. It works as a sweetener, providing about 60 percent of the sweetness of sugar. It has a lower glycemic impact compared to regular sugar, making it suitable for reduced-sugar products.
It also acts as a humectant, helping baked goods retain moisture and extend shelf life. Products such as cakes, cookies, energy bars, dried fruits, and candies often contain glycerine to improve texture and prevent dryness.
Additionally, glycerine helps dissolve flavors and colorings in beverages and confections.
Skincare and Personal Care
Glycerine is extensively utilized in moisturizers, serums, lotions, shampoos, and body washes due to its exceptional humectant properties. It effectively draws moisture from the surrounding environment into the skin’s outer layer, promoting sustained hydration and reinforcing the skin’s natural barrier.
Additionally, glycerine functions as an emollient, helping to smooth rough skin and enhance its softness. Due to its gentle nature, glycerine is well-tolerated by a wide range of skin types, including sensitive skin.
In anti-aging formulations, glycerine plays a crucial role in boosting skin hydration, which can diminish the appearance of fine lines and wrinkles that are typically caused by dehydration.
Pharmaceutical Uses
In the pharmaceutical industry, glycerine enhances the taste and texture of liquid medications, such as cough syrups, making them more palatable.
It also serves as a solvent, stabilizer, and preservative in a wide range of formulations. Additionally, glycerine-based hydrogels are commonly used in wound care to maintain moisture at the wound site and minimize irritation when dressings are removed.
Industrial Applications
Glycerine is incorporated into antifreeze formulations due to its ability to lower the freezing point of water when mixed in specific concentrations.
Additionally, it helps prevent corrosion, enhances heat transfer in cooling systems, and reduces the accumulation of rust and scale in engines and industrial equipment.
Read More: 5 Ingredients That Extend the Shelf Life of Bread
Is Glycerine Safe to Use?
Glycerine is broadly recognized as safe when used in accordance with established manufacturing practices. Regulatory bodies, including the U.S. Food and Drug Administration, deem glycerine safe for inclusion in food, cosmetics, and pharmaceutical products.
In rare instances, some individuals may experience mild skin irritation or allergic reactions. If redness or itching develops, it is advised to discontinue use and consult a healthcare professional.
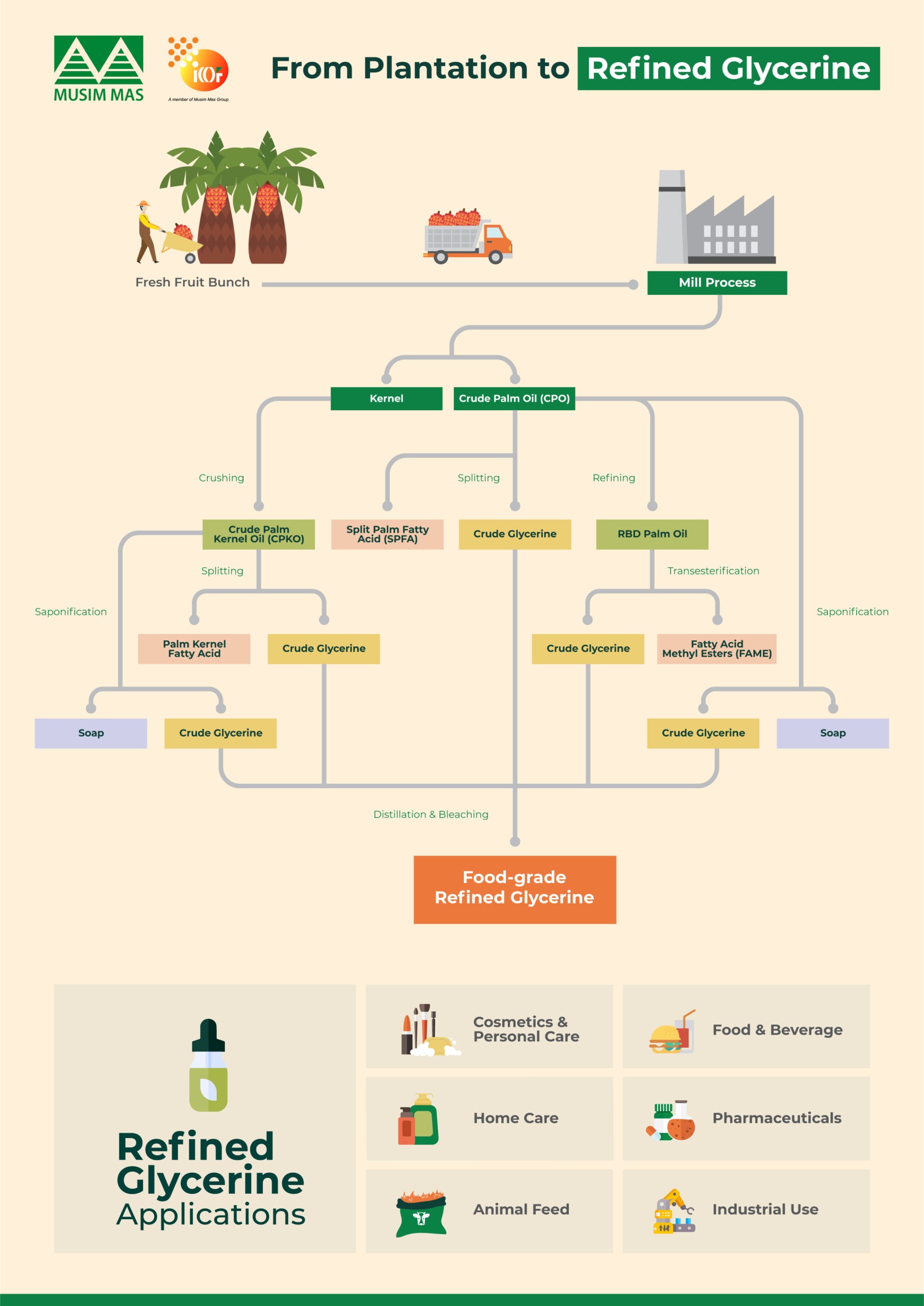
How Is Refined Glycerine Made From Palm Oil?
Palm oil and palm kernel oil serve as key raw materials for the production of vegetable-based glycerine.
The process begins with the harvesting of fresh fruit bunches from oil palm plantations, which are then processed into crude palm oil and crude palm kernel oil.
Crude glycerine can be produced through the following methods:
- Hydrolysis of palm kernel oil
- Hydrolysis of palm oil
Transesterification during biodiesel production
The resulting crude glycerine undergoes refining processes, including distillation and purification, to achieve high purity levels suitable for global markets.
Refining facilities located in Southeast Asia and Europe provide refined glycerine to manufacturers in the food, pharmaceutical, and personal care industries worldwide.
Frequently Asked Questions About Glycerine
1. Is glycerine the same as glycerol?
Yes. Glycerine and glycerol refer to the same compound. Glycerol is the more technically precise term, while glycerine is commonly used in commercial and consumer contexts. For businesses in manufacturing, the distinction does not affect the properties of the compound used in production.
2. Is glycerine safe for skin?
For the majority of individuals, glycerine is safe and beneficial for use in skincare formulations. Its ability to attract moisture and enhance the skin barrier makes it valuable in products designed for hydration and protection. Companies should consider testing products for compatibility when targeting sensitive skin to ensure the formulation is suitable and minimize the risk of irritation.
3. Is vegetable glycerine better than synthetic glycerine?
While chemically identical, the key difference between vegetable glycerine and synthetic glycerine lies in the source. Vegetable glycerine, derived from plant oils, is preferred in products marketed as natural, vegan, or eco-friendly. Manufacturers focused on these aspects should prioritize vegetable glycerine, particularly in food, cosmetic, and personal care products.
4. Why is glycerine used in food products?
Glycerine serves multiple roles in food manufacturing. It improves texture, helps retain moisture, acts as a mild sweetener, and extends the shelf life of various food products. Its versatility is particularly useful in baked goods, candies, and sugar-reduced formulations, making it an essential ingredient for food manufacturers aiming to enhance product quality and stability.
5. Does glycerine come from palm oil?
Yes, glycerine can be derived from palm oil and palm kernel oil. It is commonly produced as a by-product during biodiesel production. Once obtained, the glycerine is refined to meet the quality standards required for use in a wide range of industrial and consumer applications. For businesses utilizing glycerine in their own production processes, sourcing from palm oil may offer both cost-effective and sustainable options, depending on the market and environmental considerations.
Dutch Glycerine Refinery
Aside from our glycerine distillation plants in Indonesia, we operate a refining unit at the Delfzijl Chemical Park in the Netherlands, providing top-quality production and logistical access to major European markets.
References:
[1]:https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4415886/#R17
[2]:https://discover.univarsolutions.com/blog/glycerin-insight-production-and-market-updates/
[3]:https://foodinsight.org/what-is-glycerin/
[4]:https://polyols-eu.org/legislation/food/
[5]:https://www.engineeringtoolbox.com/glycerine-boiling-freezing-points-d_1590.html
[7]:https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4810294/